#75 GMP at the Right Level – How Smaller Companies Build Quality for Growth

The road to an eQMS

How do you go from good intentions to a functional, well-established eQMS? What does it take to build a solution that not only complies with regulations but also works in everyday life? Choosing a system is just the beginning There is a lot of focus on choosing the right platform, which is of course important. But many projects get stuck precisely […]
The road to an eQMS

How do you go from good intentions to a functional, well-established eQMS? What does it take to build a solution that not only complies with regulations but also works in everyday life? Choosing a system is just the beginning There is a lot of focus on choosing the right platform, which is of course important. But many projects get stuck precisely […]
Medical information systems in transition

With the rise of digitalization, medical information systems have become key components of healthcare infrastructure. These systems often manage sensitive patient data, control decision support and integrate with everything from medical record systems to national e-health services. At the same time, the requirements for how the systems are to be developed, managed and documented are tightening, not least through new regulations such as the Swedish Medical Products Agency's regulations on national medical information systems (NMI) and the [...]
The future of medical device manufacturing
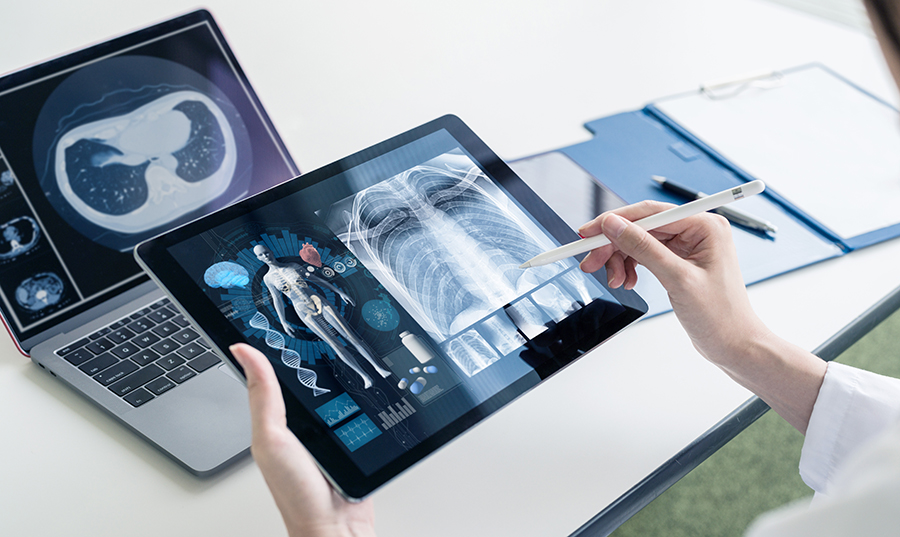
Driving innovation - but not at the expense of patient safety Innovation in Medtech is happening at a rapid pace. AI enables predictive maintenance in manufacturing supply chains, faster diagnoses and individualized treatments. But the impact of AI is not limited to this. The development process, documentation and even testing of software is faster today compared to before AI. AI is being linked to [...].
Clinical development of medical devices
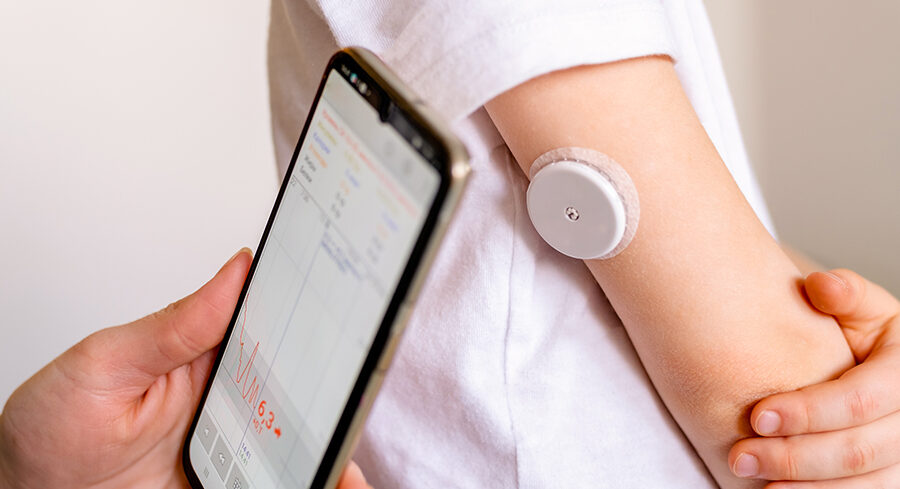
Clinical evidence is crucial, not only for the safety of patients, but for the success of the product on the market. Having a clear clinical strategy can be crucial to shortening the time from developing a product to bringing it to market - and thus reducing the risk of costly delays. "Many start too late. The clinical development should [...]
New reporting requirements for medtech products

What do the changes mean? The Swedish Medical Products Agency has clarified that all economic operators supplying medical devices on the Swedish market are affected, but only manufacturers (or if there is an authorized representative) must report deficiencies and interruptions in supply to the Agency. Others are responsible for passing on the information in the supply chain This obligation is regulated in Article 10a of the MDR and [...].
Case: Svar Life Science/Wieslab

The key to success in Life Science Building and managing a comprehensive platform offering diagnostic, pharmaceutical and Contract Research Organization (CRO) services to Life Science customers with high quality and regulatory approved systems is a challenge that requires long-term planning and technical expertise. For Svar Life Science AB, a leading Life Science company based in Skåne, Sweden, [...]
#57 QA Forum 2025 - Highlights, insights & inspiring interviews from the event with today's speakers

From industry to innovation

It's no secret that the pharmaceutical industry is regulated and complex - but it's also one of its greatest strengths. For automation engineers used to traditional process industries, entering the life sciences sector can feel like a big step. At the same time, many testify that it is precisely the structure, clarity and meaningful purpose [...]
Case: Hermes Medical Solutions' path to stronger cybersecurity
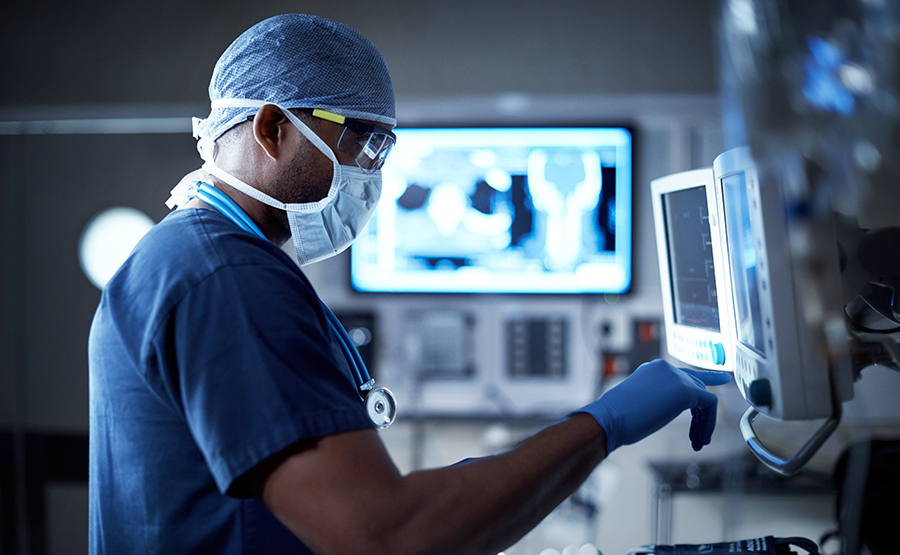
With the new NIS2 directive on the horizon, HMS was one of many companies in need of a clear picture of how its current cybersecurity efforts aligned with the new directive. The need for a clear direction With the NIS2 directive placing higher demands on information and cybersecurity, HMS wanted to identify its current [...]
Inspections and quality systems

Our subject matter experts Jessica Kjellberg and Katarina Stojanovic share their experience and insights in the field. What is a quality system and why is it important? A quality system serves as a framework to ensure that products and services meet high quality standards and are safe for use. "Whether it is GMP, Medtech, research
#54 The chicken and the egg - Introduction of an electronic testing tool for GMP

Ensuring quality & compliance with internal audits

Regulations and standards are becoming increasingly complex Companies in the life sciences, Medtech and healthcare sectors face a growing number of regulatory requirements. Standards such as ISO 13485, ISO 15189 and GMP set the bar high, and non-compliance can have far-reaching consequences - from losing certifications to affecting customer confidence and market position. Despite this, the [...]
#53 Basic insights into quality systems and inspections in Life Science

Medical Device Software

What is Medical Device Software and why is it important? Medical Device Software is software that is either a stand-alone medical device or part of a medical device, such as an app for patient monitoring or an algorithm for image analysis in diagnostics. As these systems have an impact on human health, they must meet strict requirements on functionality, safety
#52 Challenges and innovation in Swedish pharmaceutical manufacturing

The threat to public health

The consequences of non-compliance The products that manage to get through the system, despite irregularities, have serious consequences. Health risks, injuries and in some cases death are the most tangible effects. This results in a loss of confidence in life science products and the industry at large. Companies face additional financial costs in the form of product recalls and legal action - an expensive [...].
Strategic regulatory plan
What is a regulatory strategy - and why is it needed? A regulatory strategy is a document that describes the requirements and regulations that apply to products in different markets from conception to post-market surveillance. It involves identifying the specific requirements and documentation needs that the product must meet in order to be sold. This is particularly important for [...].
Case: From startup to established company - Leyr

This is a story of a young company that, thanks to its aggressive start-up approach with early intervention and focus on the right things, was able to put all its energy into developing a winning product and embedding a sustainable, long-term growth strategy throughout the business. Let us tell you more about our partnership with Leyr. A platform for [...]